Survival by Desiccation in Bdelloid Rotifers
by Ben Jeffery
BIOL/WATER 361, Fall 2013
Key taxon:
Rotifera,
Bdelloidea
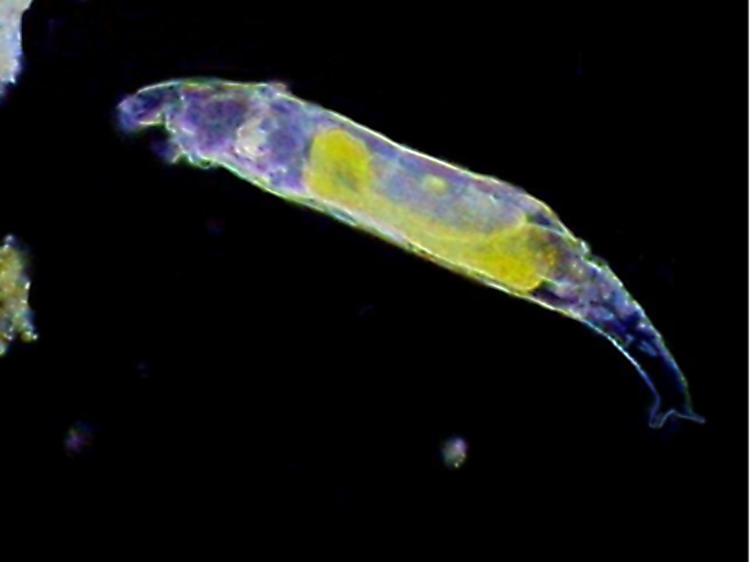 For millions of years, modes of reproduction have perpetuated life through a constantly changing environment. These modes of reproduction that allow sustainability of a species range from asexual means such as fragmentation or budding in the phylum Porifera, to complex sexual processes developed by members of the phylum Chordata. Complexity of reproductive modes aside, many species adapted multiple ways to produce offspring. As the simplest animals evolved, differences in reproductive strategies became apparent while proving to be equally effective in allowing species survival. Sexual reproduction occasionally paired with asexual reproduction was constant in early life. Species that reproduce solely by asexual means generally lack the genetic diversity to adapt, but rotifers belonging to the class Bdelloidea have survived millions of years asexually due to an ability to survive long periods of dryness through desiccation. For millions of years, modes of reproduction have perpetuated life through a constantly changing environment. These modes of reproduction that allow sustainability of a species range from asexual means such as fragmentation or budding in the phylum Porifera, to complex sexual processes developed by members of the phylum Chordata. Complexity of reproductive modes aside, many species adapted multiple ways to produce offspring. As the simplest animals evolved, differences in reproductive strategies became apparent while proving to be equally effective in allowing species survival. Sexual reproduction occasionally paired with asexual reproduction was constant in early life. Species that reproduce solely by asexual means generally lack the genetic diversity to adapt, but rotifers belonging to the class Bdelloidea have survived millions of years asexually due to an ability to survive long periods of dryness through desiccation.
Phylum Rotifera is composed of bilaterally symmetrical pseudocoelomates that inhabit both fresh and marine waters. Rotifers share many physical characteristics. Both sedentary and sessile species within the phylum possess smooth and longitudinal muscles for body movement, which provide a noticeable amount of flexibility for feeding. With a crown-like structure near the head known as the corona, rotifers typically feed on microscopic detritus suspended in the water. Digestion continues through a one-way gut supporting an anterior mouth and posterior anus. The gut includes a cilia lining to transport food to a stomach for extra cellular digestion. A similarity within phylum rotifer also includes the importance of diffusion for gas exchange with the utilization of protonephridia in waste secretion along with salt and water balances with the surrounding environment. Sharing this common structure creates the framework that defines the phylum Rotifera.
The two freshwater classes of the phylum Rotifera, Monogononta and Bdelloidea, are distinguished largely by asexual and sexual reproduction. The class Bdelloidea was taxonomically branched due to entirely reproducing through parthenogenesis, while Monogononta reproduces both sexually and asexually. Parthenogenesis, an asexual reproduction method in which females produce eggs through oocytes, occurs in bdelloid rotifers without any male representation within the class (Welch & Meselson, 2000). The eggs from an amictic female produce a clone of the parent rotifer. Asexual reproduction requires 50% of the energy needed to produce offspring when compared to sexual reproduction but sacrifices genetic variation due to the elimination of gene variation supplied by two parents (Wilson & Sherman, 2010). All gene mutations in asexual reproduction, beneficial and detrimental, are passed to the offspring, whereas in sexual reproduction, gene mutations showing a negative effect on an individual are selected against by the inability of that individual to produce numerous offspring. Overtime, sexual reproduction shapes a population with ability for adaptation. As exclusive asexual animals arise, they rarely persist for long periods of time (Welch et al 2000). This idea known as the Red Queen hypothesis is supported throughout the animal kingdom (Wilson & Sherman, 2010). The asexually persistent class Bdelloidea is a true anomaly in reproduction methods of existing animals. The taxonomic division between rotifers indicates not only the difference in reproduction, but a unique method in generating successful offspring.
Rotifers belonging to the class Bdelloidea excel in both asexual dependence and proficiency of existence in comparison to species similar in reproduction methods due to the ability of desiccation. Desiccation allows bdelloid rotifers to pause and resume living without greatly affecting their life cycle during unfavorable environmental conditions. Asexual reproduction in bdelloid rotifers typically begins in the summer with the adult female cloning daughters. In a situation posing threatening living conditions, a rotifer entering a period of desiccation can resume living once conditions improve. After continuation of the life cycle, the bdelloid rotifer can reproduce as effectively as it would have without the occurrence of desiccation (Ricci et al., 1987). For any living individual, adaptations must be made to survive changing conditions. Similar to hibernation or torpor in sexual species, desiccation in rotifers provides a successful alternative to developing genetic adaptations to handle harsher environmental situations. Because rotifers inhabit ephemeral habitats, this ability to pause and continue the life cycle when conditions are favorable separates bdelloid rotifers from previous asexual design failures in the evolutionary process (Ricci & Perletti, 2006). Desiccation in bdelloids allows the class to steadily produce viable offspring in life supporting conditions without depending on any mode other than asexual reproduction.
As desiccation provides bdelloid rotifers an escape from an unsuitable environment which would ultimately eliminate them, it also provides a survival method countering low food availability. When shortage of food occurs in any species, energy intake becomes limited which then limits the ability to make any action that requires energy expenditure. Because reproduction is energy dependent, starvation tends to lead to a priority of survival rather than production of offspring. During times of food scarcity, bdelloid rotifers enter desiccation to slow metabolic rates and, therefore, the expense of energy. Survival rates and fecundity in bdelloid rotifers after varying degrees of starvation show little detriment independent from the period of dormancy (Ricci & Perletti, 2006). Bdelloid rotifers’ ability to once again pause their life cycle during times stress allows them to survive long periods of starvation without affecting fecundity rates, lifespan, and the overall persistence within the environment. Rather than selecting for viable modes of evacuation from food-scarce environments, desiccation acts as an evolutionary counter to migration or other movement patterns observed in sexual species.
 Desiccation not only solves mortality and fecundity declines that manifest from unsuitable habitat and food-scarcity but from parasites as well. Parallel to developing genetic resistance or immune systems resist parasitism, the class Bdelloidea separates themselves from these threats by desiccation. Parasites affecting bdelloid rotifers have been identified as oomycete or hyphomycete fungi. Most of the fungi lethal to members of the class Bdelloidea belong to the genus Rotiferophthora. The fungal endoparasites exclusive to bdelloid rotifers spread infection through spores known as conidia. Once the spore enters into the rotifer, assimilative hyphae are produced in the pharynx leading to death and digestion of the rotifer. After digestion, the hyphae produce conidiophores which restart the cycle by carrying new conidia by the hundreds (Wilson & Sherman, 2010). Once again, asexual reproduction provides little answers to the arms race created between parasites and hosts due to the lack of genetic variability between generations. In cases of infected populations of bdelloid rotifers, desiccation proved to be the lifesaving alternative to sexual reproduction. The longer bdelloid rotifers stay in a state of desiccation, the greater the survivability from the parasitic genus Rotiferophthora. Not only do the rotifers outlast the fungi in a dry environment, but the rotifers also have the ability to escape the area contaminated with conidia (Wilson & Sherman, 2010). Due to the microscopic size of an individual rotifer, once in a state of desiccation, wind dispersal becomes an invaluable asset to the survival from parasites (Wilson, 2011). Through desiccation, bdelloid rotifers are able to replace natural selection for parasitic resistance with simply outlasting or outmaneuvering the genus Rotiferophthora. Desiccation not only solves mortality and fecundity declines that manifest from unsuitable habitat and food-scarcity but from parasites as well. Parallel to developing genetic resistance or immune systems resist parasitism, the class Bdelloidea separates themselves from these threats by desiccation. Parasites affecting bdelloid rotifers have been identified as oomycete or hyphomycete fungi. Most of the fungi lethal to members of the class Bdelloidea belong to the genus Rotiferophthora. The fungal endoparasites exclusive to bdelloid rotifers spread infection through spores known as conidia. Once the spore enters into the rotifer, assimilative hyphae are produced in the pharynx leading to death and digestion of the rotifer. After digestion, the hyphae produce conidiophores which restart the cycle by carrying new conidia by the hundreds (Wilson & Sherman, 2010). Once again, asexual reproduction provides little answers to the arms race created between parasites and hosts due to the lack of genetic variability between generations. In cases of infected populations of bdelloid rotifers, desiccation proved to be the lifesaving alternative to sexual reproduction. The longer bdelloid rotifers stay in a state of desiccation, the greater the survivability from the parasitic genus Rotiferophthora. Not only do the rotifers outlast the fungi in a dry environment, but the rotifers also have the ability to escape the area contaminated with conidia (Wilson & Sherman, 2010). Due to the microscopic size of an individual rotifer, once in a state of desiccation, wind dispersal becomes an invaluable asset to the survival from parasites (Wilson, 2011). Through desiccation, bdelloid rotifers are able to replace natural selection for parasitic resistance with simply outlasting or outmaneuvering the genus Rotiferophthora.
Through desiccation, the class bdelloidea continues to overcome obstacles presented by the natural world. Not only have bdelloid rotifers managed to survive millions of years solely by asexual reproduction through the aid of desiccation, but new species continue to be discovered. A carnivorous rotifer, Abrochtha carnivore, was recently discovered in Barbados as the first carnivorous rotifer within the class Bdelloidea. Abrochtha carnivore retains the typical anatomy present in bdelloid rotifers despite its carnivorous nature and slightly larger body size, including the ciliated corona used to capture prey. Though the discovered species is the first carnivore within the class, rotifers of the class Monogononta exhibit diversity in carnivorous feeding. The development of carnivorous behavior among monogononts possibly resulted from differences in reproductive modes. The selection of beneficial gene mutations within a species utilizing sexual reproduction supports the idea of quicker advancement and speciation derived from a phylogenetic ancestor. Discovery of Abrochtha carnivore supports the continued survival and evolution within the class Bdelloidea. With the divergence from the omnivorous normality, discovery of this new species not only illustrates the essential partnership between asexual reproduction and desiccation but a proven ability for bdelloid rotifers to excel in both omnivorous and carnivorous diets similar to species without sexual reproduction (Ricci et al., 2001).
In conclusion, the ability of asexual bdelloid rotifers to enter desiccation allows them to survive harsh environmental situations, detrimental parasitic fungi, as well as shortened food supplies that species reproducing sexually have evolved to overcome through natural selection. The ability of bdelloid rotifers to enter dormancy without affecting life expectancy is the sole reason this class has been able to survive successfully exclusively by asexual reproduction. As harsher environments arise, adaptations can be seen in animals to overcome and ultimately survive. Climate change is an ever-occurring driving force in environmental changes which presents new challenges to a population. The unique development of desiccation naturally paired with parthenogenesis illustrates the diversity of survival modes in invertebrates and multi-celled animals as a whole. The future provides the ability to discover new modes of survival with the persistence of those that succeeded in the past.
References Cited
- Ricci C., G. Melone & E.J. Walsh. 2001. A carnivorous bdelloid rotifer, Abrochtha carnivore n. sp. Invertebrate Biology 120: 136-41.
- Ricci C. & F. Perletti. 2006. Starve and Survive: Stress tolerance and life-history traits of a bdelloid rotifer. Functional Ecology 20: 340-346.
- Ricci C., L. Vaghi & M.L. Manzini. 1987. Desiccation of rotifers (Macrotrachela Quadricornifera): Survival and Reproduction. Ecology 68: 1488-149.
- Welch D.M. & M. Meselson. 2000. Evidence for the evolution of bdelloid rotifers without sexual reproduction or genetic exchange. Science 288: 1211-1215.
- Wilson C.G. 2011. Desiccation-tolerance in bdelloid rotifers facilitates spatiotemporal escape from multiple species of parasitic fungi. Biological Journal of the Linnaean Society 104: 564-574.
- Wilson C.G. & P.W. Sherman. 2010. Anciently asexual bdelloid rotifers escape lethal fungal parasites by drying up and blowing away. Science 327: 574.
|